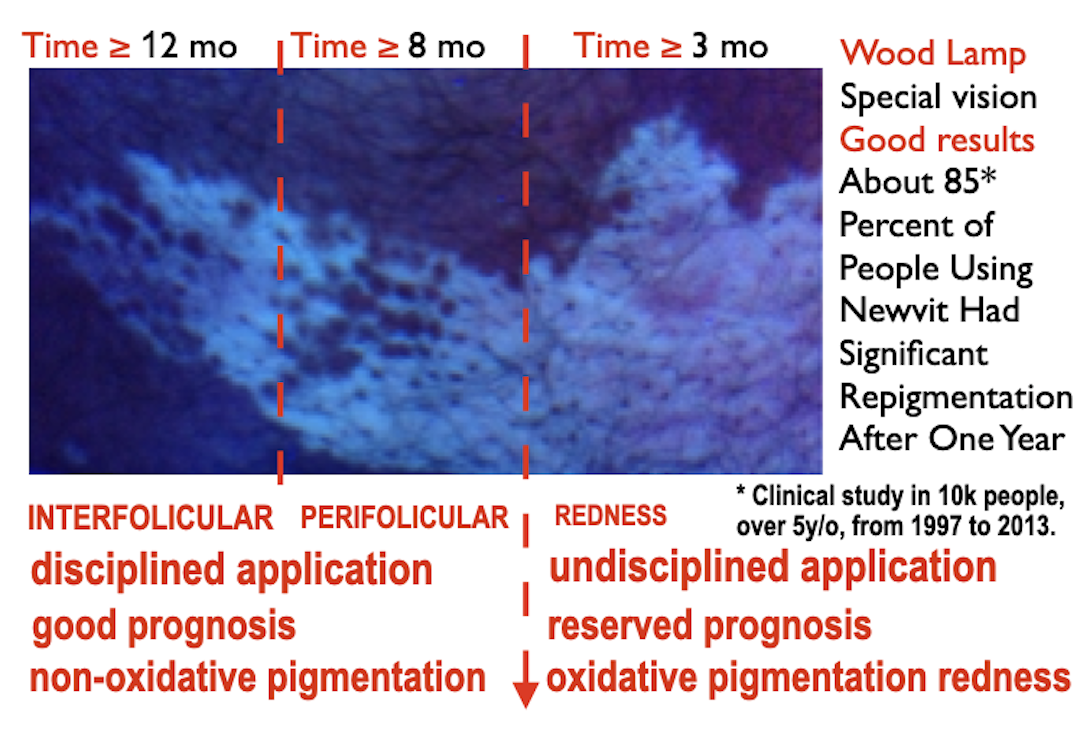
About 85 Percent of People Using Newvit Had Significant Repigmentation After One Year.
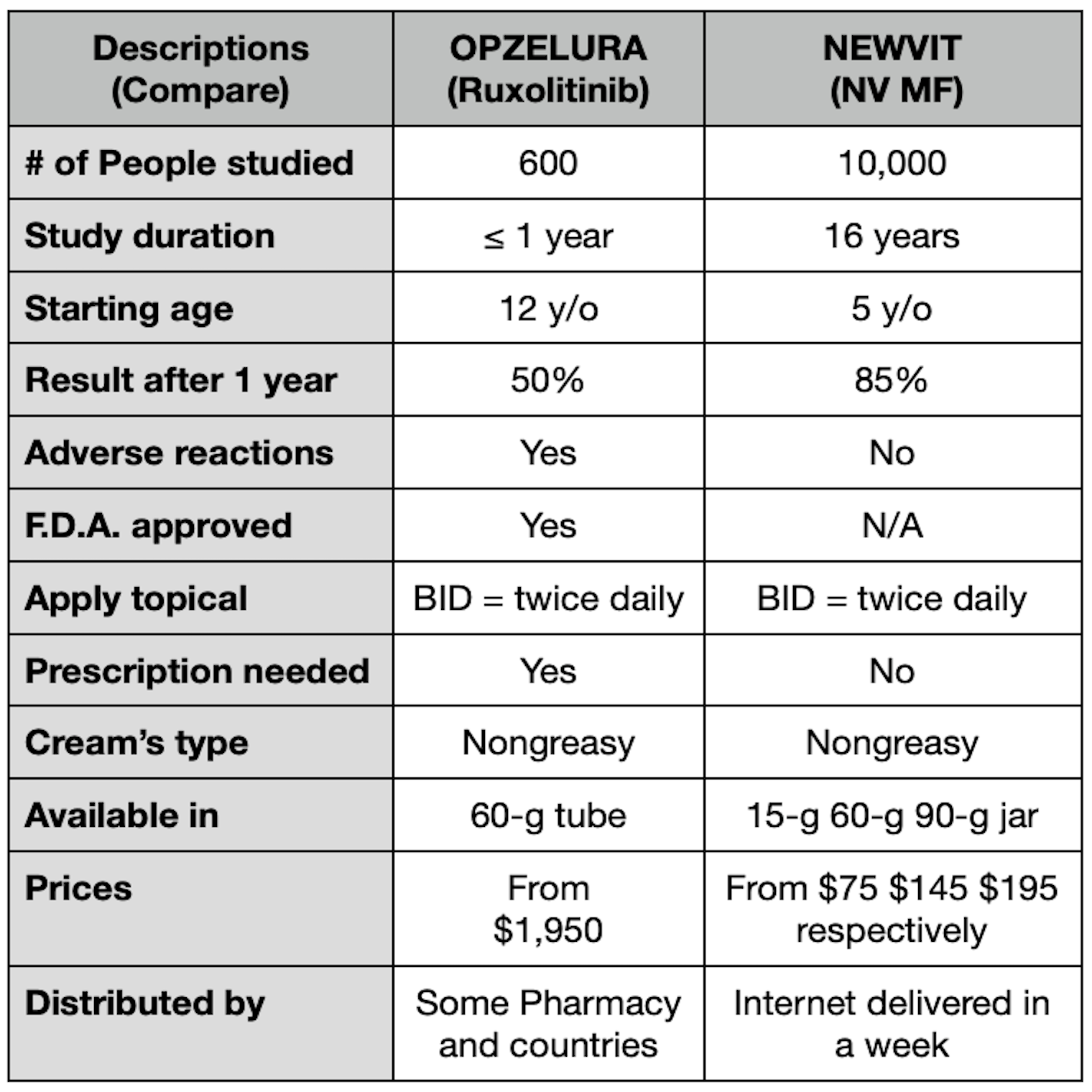
Newvit belongs to a class of cream called A Magistral Formulation (MF). Its Ingredients include: Propylene Glycol, hydroxypropyl cellulose, mannitol, magnesium stearate, talc, Calcium Acetate, Cholecalciferol, Dextrin , Glycerin, Isopropyl Palmate, Purified water, Retinyl Palmitate, Sodium Cetearyl, Tocopherol. They achieve rapid and deep absorption in the epidermis to become in a great Cells Signal Enhanced, which prevent the alterations that accompany the whitish macules in Vitiligo. Doctors prescribe Tacrolimus, Immuno modulators, Eucrisa, Ruxolitinib for topical use to treat Vitiligo. Newvit is very helpful in preventing those whitish spots from growing and prevents new spots from appearing. Its rapid absorption allows it to act faster than other dedicated products for the same purpose. Its price is affordable, it's stable for several days, and it ships internationally for delivered within a week.
Researchers have deciphered the code in the DNA that develops the alterations in some people with Vitiligo
. . CGA TAT TCC CAT CGA TC. . (L155H A>T 17p13.2)
How is the newvit applied?
People using Newvit apply the cream twice daily to the affected areas, covering up to 5 percent of their body’s surface area. It may take 20 weeks or more for people with vitiligo to see satisfactory results, according to the studies that allowed to develop this magistral formulation.
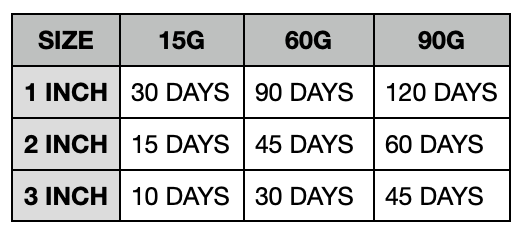
People apply as a thin layer, twice daily to affected skin on up to 5% BSA (no more than 60 g per week).
For topical use only. Not for ophthalmic, oral, or intravaginal use. Stop using if appear signs and symptoms (eg, itch, rash, and redness) in the skin. If signs and symptoms do not improve within 8 weeks, patients should be reexamined by their healthcare provider.
BID=twice daily; BSA=body surface area.
What studies have been done?
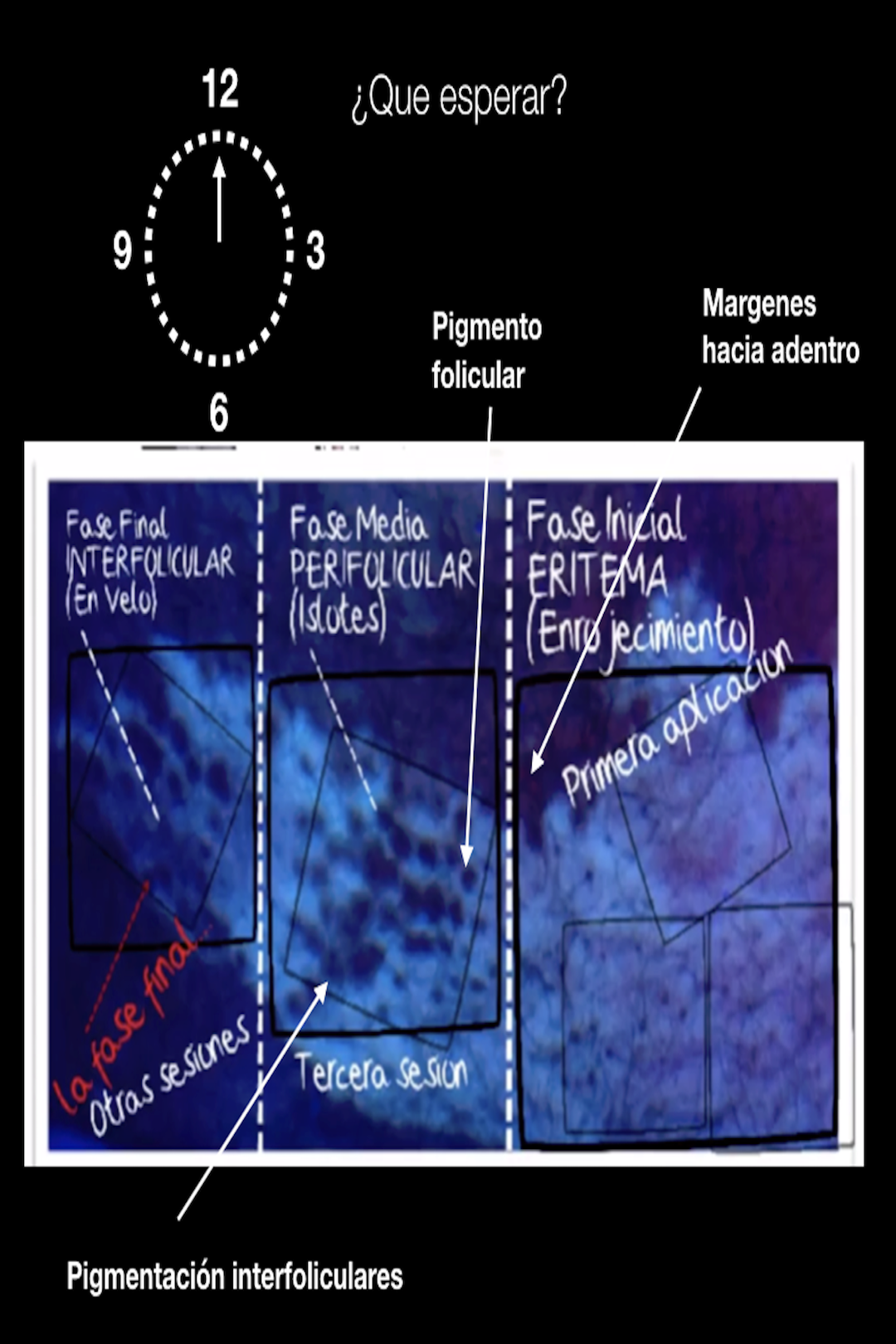
From 1997 to 2013, clinical studies were carried out that included more than 10,000 people over 5 years of age, supervised by specialist doctors who monitored with special vision means, dermoscope and wood lamp examinations, the edges, the presence of pigmentation around the hairs, the pigmentation between each hair on the skin, as well as the absence of skin atrophy, formation of new visible capillaries, or any undesirable signs. People had to attend their evaluation weekly or monthly. The results obtained mostly depended on the discipline in which the Newvit cream was used. Many of those who showed great interest and had the resources to treat themselves had favorable changes from the first 12 weeks. The vast majority saw better results after 20 weeks of continuous application. Another great finding is that no one except a single 13-year-old showed signs of hypersensitivity in their lips and was considered to be allergic to one of the ingredients. Its use was avoided in her, and topical hydrocortisone and loratadine 10 mg were applied for 30 days. No other person complained of any discomfort or unwanted side effects were seen. Why has Newvit not been evaluated by the F.D.A.? Are compounded medications FDA-approved? Compounded drugs are not FDA-approved. This means that FDA does not verify the safety or effectiveness of compounded drugs. Consumers and health professionals rely on the drug approval process for verification of safety, effectiveness, and quality. Newvit has been created by people with academic medical training, who without resources have made an effort to achieve better results at a global level, without so many limitations and with the resources of those interested. As it is a master formulation that must be modified according to the results obtained, the F.D.A. does not review these necessary changes and it is not within its functions to approve them although many of the products used for the formula enjoy its approval, not all of them have been evaluated by the F.D.A.